U petnaestogodišnjoj studiji, koja je ispitala 174.505 stanovnika područja Nagano u Japanu, utvrđeno je da uzgajivači gljiva imaju mnogo manju stopu smrtnosti od raka nego populacija u celini (97,1 / 100.000 u poređenju sa 160,1 / 100.000).
IkekawaT. Cancer Risk Reduction by Intake of Medicinal Mushrooms. IntJ Med Mush. 2005;7(3):347
Epidemiološka studija koja je obuhvatala 2.000 kineskih žena, polovinu sa rakom dojke i polovinu bez, našla je smanjenje rizika od raka dojke kod žena koje su redovno konzumirale gljive (10 grama svežih ili 4 grama sušenih gljiva dnevno).
To investigate effects of dietary mushrooms and joint effects of mushrooms and green tea on breast cancer, a case-control study was conducted in southeast China in 2004-2005. The incident cases were 1,009 female patients aged 20-87 years with histologically confirmed breast cancer. The 1,009 age-matched controls were healthy women randomly recruited from outpatient breast clinics. Information on frequency and quantity of dietary intake of mushrooms and tea consumption, usual diet, and lifestyle were collected by face-to-face interview using a validated and reliable questionnaire. Compared with nonconsumers, the Odds ratios (Ors) were 0.36 (95% CI = 0.25-0.51) and 0.53 (0.38-0.73) for daily intake of >or=10 g fresh mushrooms and >or=4 g dried mushrooms, based on multivariate logistic regression analysis adjusting for established and potential confounders. There were dose-response relationships with significant tests for trend (p less than 0.001). The inverse association was found in both pre- and postmenopausal women. Compared with those who consumed neither mushrooms nor green tea, the ORs were 0.11 (0.06-0.20) and 0.18 (0.11-0.29) for daily high intake of fresh and dried mushrooms combined with consuming beverages made from >or=1.05 g dried green tea leaves per day. The corresponding linear trends were statistically significant for joint effect (p less than 0.001). We conclude that higher dietary intake of mushrooms decreased breast cancer risk in pre- and postmenopausal Chinese women and an additional decreased risk of breast cancer from joint effect of mushrooms and green tea was observed. More research is warranted to examine the effects of dietary mushrooms and mechanism of joint effects of phytochemicals on breast cancer.
Dve korejske studije, upoređujući žene sa histološki potvrđenim rakom dojke i isti broj žena bez raka dojke, pronašle su jaku obrnutu korelaciju između potrošnje gljiva i rizika od ovog oblika kancera.
To evaluate the association between dietary mushroom intake and breast cancer risk, a total of 362 women between the ages of 30 and 65 years who were histologically confirmed to have breast cancer were matched to controls by age (+/-2 years) and menopausal status. Mushroom intake was measured via a food frequency questionnaire that was administered by well-trained interviewers. The associations between the daily intake and the average consumption frequency of mushrooms with breast cancer risk were evaluated using matched data analysis. Both the daily intake (5th vs. 1st quintile, OR = 0.48, 95% CI = 0.30-0.78, p for trend 0.030) and the average consumption frequency of mushrooms (4th vs. 1st quartile, OR = 0.54, 95% CI = 0.35-0.82, p for trend 0.008) were inversely associated with breast cancer risk after adjustment for education, family history of breast cancer, regular exercise [>or=22.5 MET (metabolic equivalent)-hr/week], BMI (body mass index, Kg/m(2)), number of children and whether they are currently smoking, drinking or using multivitamin supplements. Further adjustments were made for energy-adjusted carbohydrate, soy protein, folate and vitamin E levels, which tended to attenuate these results. After a stratification was performed according to menopausal status, a strong inverse association was found in postmenopausal women (OR = 0.16, 95% CI = 0.04-0.54, p for trend = 0.0058 for daily intake; OR = 0.17, 95% CI = 0.05-0.54, p for trend = 0.0037 for average frequency), but not in premenopausal women. In conclusion, the consumption of dietary mushrooms may decrease breast cancer risk in postmenopausal women.

Antikancerogeni lekovi na bazi medicinskih gljiva registrovani u Japanu
| Krestin / PSK (oralno) | Lentinan (injekcija) | Sizofran / SPG (injekcija) |
| Trametes versicolor (Coriolus) micelijum | Lentinula edodes (Šitake) telo gljive | Schizophillan commune središnji deo kulture |
| maj 1977. godine | decembar 1985. godine | januar 1986. godine |
| Rak želuca, pluća i dojke | Rak želuca | Rak grlića materice |
Coriolus Versicolor
Kliničke studije o dejstvu PSK na rak želuca
| Rak želuca Stadijum IV | Kaibara et al, 1976 Operacija + MCC +/- PSK sa hemoterapijom | 66 | PSK sa hemoterapijom – udvostručeno dvogodišnje preživljavanje |
| Uznapredovan rak želuca sa metastazama | Kodama et al, 1982 Operacija + MCC +/- PSK sa hemoterapijom | 450 | PSK sa hemoterapijom – udvostručeno petogodišnje preživljavanje |
| Rak želuca, Stadijum III (dvostruko slepo istraživanje) | Kondo i Torisu, 1985 Operacija bez hemoterapije +/- PSK | 144 | PSK produžio period bez bolesti i poboljšao imunitet |
| Uznapredovan rak želuca sa metastazama | Maehara et al, 1990 Operacija + MCC +/- PSK sa hemoterapijom | 255 | PSK sa hemoterapijom – produženo petnaestogodišnje preživljavanje |
| Rak želuca I-IV | Nakazato et al, 1994 Operacija +/- PSK sa hemoterapijom | 253 | PSK produžio petogodišnje preživljavanje i period bez bolesti |
Immunoceuticals can be considered as substances having immunotherapeutic efficacy when taken orally. More than 50 mushroom species have yielded potential immunoceuticals that exhibit anticancer activity in vitro or in animal models and of these, six have been investigated in human cancers. All are non-toxic and very well tolerated. Lentinan and schizophyllan have little oral activity. Active Hexose Correlated Compound (AHCC) is poorly defined but has shown early clinical promise. Maitake D-Fraction has limited proof of clinical efficacy to date, but controlled research is underway. Two proteoglycans from Coriolus versicolor – PSK (Polysaccharide-K) and PSP (Polysaccharide-Peptide – have demonstrated the most promise. In Japanese trials since 1970, PSK significantly extended survival at five years or beyond in cancers of the stomach, colon-rectum, esophagus, nasopharynx, and lung (non-small cell types), and in a HLA B40-positive breast cancer subset. PSP was subjected to Phase II and Phase III trials in China. In double-blind trials, PSP significantly extended five-year survival in esophageal cancer. PSP significantly improved quality of life, provided substantial pain relief, and enhanced immune status in 70-97 percent of patients with cancers of the stomach, esophagus, lung, ovary, and cervix. PSK and PSP boosted immune cell production, ameliorated chemotherapy symptoms, and enhanced tumor infiltration by dendritic and cytotoxic T-cells. Their extremely high tolerability, proven benefits to survival and quality of life, and compatibility with chemotherapy and radiation therapy makes them well suited for cancer management regimens.
Kliničke studije o dejstvu PSK – drugi kanceri
| Rak debelog creva (dvostruko slepo istraživanje) | Kondo i Torisu, 1990 Operacija bez hemoterapije +/- PSK | 110 | PSK produžio osmogodišnje preživljavanje i period bez bolesti |
| Rak jednjaka | Ogoši et al, 1995 Operacija + radioterapija +/- hemoterapija +/- PSK | 158 | PSK produžio petogodišnje preživljavanje posle operacije + RT + CT i normalizovao faktore seruma |
| Rak pluća, Stadijumi I-III | Hajakava et al, 1993 Radioterapija +/- PSK | 185 | PSK produžio petogodišnje preživljavanje 2-4 puta u svim stadijumima. 70 godina maksimalne koristi |
| Rak dojke, ER +/-, Stadijum II, postoperativni | Toi et al, 1992 MMC +/- Tamoxifen +/- Ftorafur +/- PSK | 914 | PSK produžio preživljavanje u ER neg, bez metastaza |
| Rak dojke, Stadijumi I-II | Iino et al, 1995 FEMP hemoterapija + Levamisol ili PSK | 227 | PSK težio ka produženju desetogodišnjeg preživljavanja i perioda bez bolesti. HLA B40+ 100% |
U sistematskoj i meta-analizi koje su prikupile i analizirale efikasnost ekstrakta Coriolusa na preživljavanje pacijenata obolelih od raka, u 13 slučajnih placebo kontrolisanih kliničkih studija utvrđeno je da Coriolus može dovesti do značajnog preživljavanja u poređenju sa standardnim uobičajenim lecenjem raka.
Kod pacijenata tretiranih Coriolusom, nastalo je 9 % apsolutnog smanjenja mortaliteta u 5 godina, što je dovelo do jednog dodatnog živog pacijenta na svakih 11 lečenih. Kod pacijenata sa karcinomom dojke, kancerom želuca ili debelog creva, koji su lečeni hemoterapijom, efekti kombinacije preparata Coriolusa na ukupnu stopu preživljavanja od 5 godina su više nego očigledni, ali ne i kod raka jednjaka i ždrela.
Analiza podgrupa nije mogla da zaključi koja vrsta tretmana protiv raka može maksimalno da iskoristi prednosti Coriolusa.
MD Anderson Cancer Center “Centar za rak Univerziteta u Teksasu” dao je sledeće naučne tvrdnje o medicinskoj gljivi Coriolus Versicolor:
- “Coriolus je kandidat koji obećava kada je u pitanju hemoprevencija zahvaljujući višestrukim efektima koje ima na maligni proces, ograničene sporedne pojave i sigurnost kod oralnih dnevnih doza tokom produženog vremena konzumiranja”.
- “Izgleda da PSK i PSP višestruko deluju na maligne procese tako što inhibiraju adheziju, invaziju, pokretljivost i metastatički rast tumorskih ćelija kod životinja”.
- “Adhezija i invazija sprečene su suzbijanjem proizvodnje ćelijskog MMP enzima od strane malignih ćelija”.
- “Pokretljivost malignih ćelija i kasnija mogućnost adhezije na krvne sudove sprečena je supresijom agregacije trombocita indukovane tumorskim ćelijama i anti-angiogenskim faktorima”.
- “Imuni odgovora domaćina ne izgleda da može biti pod uticajem PSK u normalnim uslovima, ali imuni sistem pod teretom tumora ili hemoterapije, vraća se u normalu pod dejstvom PSK u ispitivanjima na životinjama”.
- “Imuna obnova uključila je antitela i proizvodnju citokina, kao i poboljšanje smanjene antitumorske aktivnosti prirodnih ćelija ubica, T ćelija, makrofaga i limfocita periferne krvi “in vivo” i “in vitro”.
- “PSK je pokazao da sprečava pad imune sposobnosti u perioperativnom periodu i inhibira rast rezidualnih tumora uz krioablaciju”.
- “PSP je pokazao da može preokrenuti tumorom indukovane imunodeficijencije kod miševa obolelih od sarkoma, povećanjem nivoa imunoglobulina G i C3. Takođe je povezan sa povećanjem belih krvnih zrnaca, CD4, CD8, B-limfocita i neutrofila, zajedno sa većom stopom preživljavanja miševa sa tumorima”.
The University of Texas, “MD Anderson Cancer Center”, Coriolus versicolor Detailed Scientific Review
Shiitake
Meta-analiza 5 kliničkih studija sa ukupno 650 učesnika pokazala je da dodavanje lentinana iz gljive Shiitake standardnoj hemioterapiji, i to svega 2 mg nedeljno, nudi značajnu prednost u odnosu na samu hemoterapiju u pogledu preživljavanja pacijenata sa naprednim kancerom želuca. Takođe, bolji rezultat pokazao se kod pacijenata sa metastazama na limfnim čvorovima u odnosu na one koje nemaju metastaze na tim mestima.
BACKGROUND:
In the present study, the effect of immunochemotherapy with lentinan compared with that of chemotherapy alone was evaluated in patients with advanced gastric cancer through individual patient data (IPD) meta-analysis.
MATERIALS AND METHODS:
Based on a computerized and manual search, all eligible centrally randomized controlled trials (RCT) which compared chemotherapy regimens with or without lentinan administration for advanced gastric cancer patients were included.
RESULTS:
In total, 650 IPD from 5 trials were available. Lentinan significantly prolonged the overall survival (stratified log-rank p=0.011). The overall hazard ratio (HR) was 0.80 (95% confidence interval=0.68-0.95) and there was no heterogeneity between trials. Additionally, lentinan was possibly more effective in patients with lymph-node metastasis than in non-node metastasis patients (P for interaction=0.077).
CONCLUSION:
The addition of lentinan to standard chemotherapy offers a significant advantage over chemotherapy alone in terms of survival for patients with advanced gastric cancer.
Dodatne naučne studije potvrđuju produzeno prezivljavanje, smanjene sporednih efekata hemoterapije i poboljšanje kvaliteta života kod pacijenata sa kolorektalnim i hepatocelularnim tumorom, rakom dojke i metastastatskim rakom prostate. U ogledu sa 69 pacijenata, sa metastatskim karcinom prostate, 50% preživljavanje u lecenoj grupi i kontrolnoj grupi bilo je 48, odnosno 35 meseci, dok je petogodišnja stopa preživljavanja kod lečenih pacijenata bila 43 % u odnosu na 29 % pacijenata iz kontrolne grupe.
In order to evaluate clinical efficacy of Lentinan (LNT), a purified polysaccharide extracted from Lentinus edodes, randomized controlled studies with envelope method have been conducted on the patients with advanced or recurrent, stomach, colo-rectal and breast cancer. Administration condition of LNT for gastrointestinal cancer was designed as the following: LNT was administered intravenously at doses of 1 mg/person/day twice a week or 2 mg/person/day once a week in combination with C + 5-FU (MF) or tegafur (FT). Control therapy was the administration of MF or FT alone. Survival curve drawn by Kaplan-Meier’s method showed that life span prolongation effect of LNT was observed with statistical significance (P less than 0.05 or P less than 0.01) by use of generalized Wilcoxon’s test. Moreover, improvement of host immune responses was observed in LNT treated group, and hematological survey showed that incidence rate of abnormal value was significantly low in LNT treated group. Thus, LNT should be effective for the patients with advanced or recurrent stomach or colo-rectal cancer in combination with chemotherapeutic agents such as MF or FT. Regarding advanced or recurrent breast cancer, study is underway. LNT has been administered as an agent for supportive therapy to the patients with complete response, partial response or stable diseases which were induced by prior surgery of oophorectomy. Again, life span prolongation effect of LNT has been observed with statistical significance (P less than 0.05). This result suggests that LNT would also be effective for the patients with advanced or recurrent breast cancer as an agent for supportive therapy.
A prospective, randomized multi-center study was conducted to assess the clinical effectiveness of Lentinan, an immunomodulatory agent, in the metastatic prostate cancer. Of seventy-five patients enrolled from July 1987 to June 1992, 69 were eligible. All patients received hormonal therapy and chemotherapy using Tegafur p.o. at a dose of 400-800 mg/day. While 33 patients received Lentinan i.m. for at least three months, the other 36 did not. The dose of Lentinan was 2 mg weekly for inpatients and 4 mg every other week for outpatients. The mean age of treated and control patients was 70 (range; 53-83) and 71 (range; 50-86), respectively. The 50% survival length of treated and control patients was 48 and 35 months, respectively. The five-year survival rate of treated patients was 43% according to the Kaplan-Meier method, while that of control patients was 29% (p less than 0.05). We conclude that Lentinan is effective in metastatic prostate cancer when incorporated into hormonochemotherapy.
Iako se obično isporučuje injekcijom, lentinan je takođe i oralno raspoloživ mada je klinička doza verovatno veća.
OBJECTIVES:
Lentinan was extracted from shiitake mushrooms (Lentinus edodes) via a new cost-effective procedure that resulted in high purity (88%) and yield. Unlike previous reports whereby the lentinan was given parenterally, in this study the emphasis was on the oral administration of lentinan. The goal is to document whether the efficacy of the antitumor property is still expressed through this route of administration.
DESIGN:
Initial study on the action of lentinan was conducted using murine lymphoma (K36) cells in a AKR mouse model. Further investigation on the effectiveness of the extracted lentinan was then performed using human colon-carcinoma cell lines in mice. Six established human colon-carcinoma cell lines segregated into three groups of different degrees of differentiation were used in this study. One group was not fed (control) and the second group was prefed with lentinan for 7 days prior to inoculations with the cancer cells. The size of the tumors that developed was rated after 1 month.
RESULTS:
Significant regression in tumor formation was observed in prefed mice compared to control (unfed) mice when K36 or human colon-carcinoma cells were used. Significant reductions in the size of the tumors were observed in mice prefed with lentinan. Follow-up investigation proceeded with the use of nude mice (athymic). Lymphocytes extracted from AKR mice prefed with lentinan for 7 days were inoculated into the nude mice. This was then followed by inoculation of the human colon-carcinoma cell lines into these mice. Much smaller tumors were formed in nude mice inoculated with lymphocytes, in contrast to the larger tumor formed in nude mice without lymphocytes inoculation.
CONCLUSION:
This study showed that the antitumor property of lentinan was maintained with oral administration. In addition, „primed“ lymphocytes, when given passively to immunodeficient mice, were able to retard the development of tumors in these mice.
Lentinan is a purified β-glucan isolated from Lentinus edodes (shiitake mushroom). Past studies have shown that lentinan possesses strong inhibitory effects on tumors induced in mice via intravenous and intraperitoneal injection. The antitumor and immunomodulation effects of lentinan through oral administration were investigated in our study, and the results obtained proved its efficacy. The results showed that prefeeding of the mice for 7 days at an optimum dose of 3 mg/mouse was most effective against tumor induction, achieving a tumor inhibition rate (TIR) of 94.44%. Four key cytokines involved in the inhibition of tumor development—namely, interleukin-1α, interleukin-2, interferon-γ, and tumor necrosis factor-α—were induced significantly in the blood of the lentinan-fed mice. This also indicated the immunomodulating effect of lentinan. Further investigation showed that lentinan is capable of activating the lymphocytes of the host. Three types of immunodeficient mice—namely, nude mice (athymic mice), B-cell-deficient mice, and SCID (severe combined immunodeficient) mice—were used to demonstrate the protective capability of the activated lymphocytes. Normal AKR mice fed with lentinan for 7 days were sacrificed, and lymphocytes were isolated from their spleens. Adoptive transfer of these lymphocytes was done by inoculation via the immunodeficient mice tails. The sizes of the developed tumors (induced using the human colon carcinoma cells) were scored after 1 month. Large tumors were observed to be present in those immunodeficient mice that received lymphocytes from buffer solution-fed mice, whereas very small tumors or no tumors were observed in mice that received lymphocytes from lentinan-fed mice. The passively transferred lentinan-primed lymphocytes were able to restrict tumor development in these immunodeficient mice.
Maitake
U studiji koja je ispitala 165 pacijenata sa dijagnozom kancera III i IV stadijuma davana im je Maitake gljiva, pri čemu je zabeležena regresija tumora i/ili značajno simptomatsko poboljšanje i to kod 66 % pacijenata sa karcinomom pluća, 54 % pacijenata sa rakom jetre, 56 % obolelih od raka pankreasa i 74 % pacijenata sa karcinomom dojke.
U odvojenoj studiji, kombinacija ekstrakata iz Maitake gljive dala je slična poboljšanja, i to kod 58,3% pacijenata obolelih od raka jetre, 68,8% obolelih od raka dojke i 62,5% obolelih od kancera pluća.
Can Maitake MD-Fraction Aid Cancer Patients? Kodama N, Komuta K, Nanba H. Alt Med Rev. v.7,3. 2002
Maitake mushroom (Grifola frondosa) MD-fraction containing beta-1,6 glucan with beta-1,3 branched chains has previously exhibited strong anticancer activity by increasing immune-competent cell activity.1,2 In this non-random case series, a combination of MD-fraction and whole maitake powder was investigated to determine its effectiveness for 22- to 57-year-old cancer patients in stages II-IV. Cancer regression or significant symptom improvement was observed in 58.3 percent of liver cancer patients, 68.8 percent of breast cancer patients, and 62.5 percent of lung cancer patients. The trial found a less than 10-20 percent improvement for leukemia, stomach cancer, and brain cancer patients. Furthermore, when maitake was taken in addition to chemotherapy, immune-competent cell activities were enhanced 1.2-1.4 times, compared with chemotherapy alone. Animal studies have supported the use of maitake MD-fraction for cancer.
Laboratorijske studije su takođe pokazale uzajamno dejstvo ekstrakta Maitake (D-frakcija) i hemoterapije (mitomicin C), kao i značajno ublažavanje neželjenih efekata hemoterapije, kao što su gubitak apetita, povraćanje, mučnina, gubitak kose i leukopenija.
Maitake extracts and their therapeutic potential -A Review. Mayell M. Alt Med Rev, v.6.1. 2001
Maitake (Grifola frondosa) is the Japanese name for an edible fungus with a large fruiting body characterized by overlapping caps. It is a premier culinary as well as medicinal mushroom. Maitake is increasingly being recognized as a potent source of polysaccharide compounds with dramatic health-promoting potential. The most recent development is the MD-fraction, a proprietary maitake extract its Japanese inventors consider to be a notable advance upon the preceding D-fraction. The D-fraction, the MD-fraction, and other extracts, often in combination with whole maitake powder, have shown particular promise as immunomodulating agents, and as an adjunct to cancer and HIV therapy. They may also provide some benefit in the treatment of hyperlipidemia, hypertension, and hepatitis.
Agaricus blazei
Ekstrakti polisaharida iz gljive Agaricus blazei pokazuju snažnu “in vitro“ i “in vivo“ aktivnost protiv niza kancerogenih ćelijskih linija, narocito kod raka pluća i jajnika.
Agaricus blazei is a medically important mushroom widely eaten and prescribed in Japan. Polysaccharide fractions were prepared from cultured A. blazei by repeated extraction with hot water (AgHWE), cold NaOH (AgCA), and then hot NaOH (AgHA). By chemical, enzymic, and NMR analyses, the primary structures of AgHWE, AgCA, and AgHA were mainly composed of 1,6-beta-glucan. Among these fractions, the NaOH extracts showed antitumor activity against the solid form of Sarcoma 180 in ICR mice. To demonstrate the active component in these fractions, several chemical and enzymic treatments were applied. These fractions were found to be i) neutral beta-glucan passing DEAE-Sephadex A-25, ii) resistant to periodate oxidation (I/B) and subsequent partial acid hydrolysis (I/B/H), iii) resistant to a 1,3-beta-glucanase, zymolyase, before I/B, but sensitive after I/B/H. In addition, after I/B/H treatment of the neutral fraction of AgCAE, a signal around 86 ppm attributable to 1,3-beta glucosidic linkage was detectable in the 13C-NMR spectrum. These facts strongly suggest that a highly branched 1,3-beta-glucan segment forms the active center of the antitumor activity.
Kliničke studije dale su pozitivne rezultate kod brojnih pacijenata prevashodno obolelih od raka dojke i to primenom Agaricusa u neodređenim dozama. Osim ovog rezultata, ekstrakt polisaharida iz ove gljive zabeležio je i povećanu aktivnost NK ćelija (ćelije ubice) i redukciju propratnih efekata hemoterapije.
Posebna studija iz 2008. godine potvrdila je povećanu aktivnost NK ćelija uz dozu ekstrakta polisaharida Agaricusa od 3 grama dnevno.
Reishi
Rezultati meta-analize 5 slučajnih kontrolisanih studija pokazali su da su pacijenti kojima je data Reishi gljiva zajedno uz hemioterapiju ili radioterapiju 1,27 puta pozitivnije odgovarali na terapiju u odnosu na one pacijente bez dodavanja gljive. Sam tretman Reishi gljivom nije pokazao istu stopu regresije koja je viđena u kombinovanoj terapiji.
Četiri studije su pokazale da su pacijenti u Reishi grupi imali relativno bolji kvalitet života u poređenju sa kontrolnim grupama. Samo jedna od studija zabeležila je minimalne sporedne efekte, poput mučnine i nesanice.
Rezultati su pokazali da su indikatori funkcije imunog sistema nagovestili da Reishi gljiva istovremeno povećava procenat CD3, CD4 i CD8.
BACKGROUND:
Ganoderma lucidum is a natural medicine that is widely used and recommended by Asian physicians and naturopaths for its supporting effects on immune system. Laboratory research and a handful of preclinical trials have suggested that G. lucidum carries promising anticancer and immunomodulatory properties. The popularity of taking G. lucidum as an alternative medicine has been increasing in cancer patients. However, there is no systematic review that has been conducted to evaluate the actual benefits of G. lucidum in cancer treatment.
OBJECTIVES:
To evaluate the clinical effects of G. lucidum on long-term survival, tumour response, host immune functions and quality of life in cancer patients, as well as adverse events associated with its use.
SEARCH METHODS:
The authors ran an extensive set of databases including the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, EMBASE, NIH, AMED, CBM, CNKI, CMCC and VIP Information/Chinese Scientific Journals Database was searched for randomised controlled trials (RCTs) in October 2011. Other strategies used were scanning the references of articles retrieved, handsearching of the International Journal of Medicinal Mushrooms and contact with herbal medicine experts and manufacturers of G. lucidum.
SELECTION CRITERIA:
To be eligible for being included in this review, studies had to be RCTs comparing the efficacy of G. lucidum medications to active or placebo control in patients with cancer that had been diagnosed by pathology. All types and stages of cancer were eligible for inclusion. Trials were not restricted on the basis of language.
DATA COLLECTION AND ANALYSIS:
Five RCTs met the inclusion criteria and were included in this review. Two independent review authors were assigned to assess the methodological quality of individual trials. Common primary outcomes were tumour response evaluated according to the World Health Organization (WHO) criteria, immune function parameters such as natural killer (NK)-cell activity and T-lymphocyte co-receptor subsets, and quality of life measured by the Karnofsky scale score. No trial had recorded long-term survival rates. Associated adverse events were reported in one study. A meta-analysis was performed to pool available data from the primary trials. Results were gauged using relative risks (RR) and standard mean differences (SMD) for dichotomous and continuous data respectively, with a 95% confidence interval (CI).
MAIN RESULTS:
The methodological quality of primary studies was generally unsatisfying and the results were reported inadequately in many aspects. Additional information was not available from primary trialists. The meta-analysis results showed that patients who had been given G. lucidum alongside with chemo/radiotherapy were more likely to respond positively compared to chemo/radiotherapy alone (RR 1.50; 95% CI 0.90 to 2.51, P = 0.02). G. lucidum treatment alone did not demonstrate the same regression rate as that seen in combined therapy. The results for host immune function indicators suggested that G. lucidum simultaneously increases the percentage of CD3, CD4 and CD8 by 3.91% (95% CI 1.92% to 5.90%, P
AUTHORS’ CONCLUSIONS:
Our review did not find sufficient evidence to justify the use of G. lucidum as a first-line treatment for cancer. It remains uncertain whether G. lucidum helps prolong long-term cancer survival. However, G. lucidum could be administered as an alternative adjunct to conventional treatment in consideration of its potential of enhancing tumour response and stimulating host immunity. G. lucidum was generally well tolerated by most participants with only a scattered number of minor adverse events. No major toxicity was observed across the studies. Although there were few reports of harmful effect of G. lucidum, the use of its extract should be judicious, especially after thorough consideration of cost-benefit and patient preference. Future studies should put emphasis on the improvement in methodological quality and further clinical research on the effect of G. lucidum on cancer long-term survival are needed. An update to this review will be performed every two years.
Ganoderinska i lucidenska kiselina
Ganoderinska i lucidenska kiselina koje se nalaze u gljivi Reishi pokazuju:
- Sedativni efekat na centralni nervni sistem
- Jaku protivupalnu aktivnost
- Inhibiciju oslobođenje histamina
- Antihepatotoksično dejstvo
- Antihipertenzivno dejstvo
- Antivirusno dejstvo
- Antitumorna aktivnost
Antitumorna aktivnost ganoderinske i lucidenske kiseline
- Suzbijanje rasta kancera i angiogeneze preko modulacije AP-1 i NF-kB signalizacije
- Indukcija apoptoze posredstvom mitohondrija
- Inhibicija invazivnosti ćelija raka
- Inhibicija aktivnosti aromataze
- Suzbijanje steroida 5 alfa-reduktaze, koji konvertuje testosteron u dihidrotestosteron (DHT), a koji igra važnu ulogu u razvoju raka prostate i benignog uvećanja prostate
- Pospešenost hemosenzitivnosti kroz inhibiciju JAK-STAT3 signalnog puta
Pilot klinička studija pokazuje da spore Reishi gljive poboljšavaju umor kod pacijenata obolelih od raka dojke, koji su podvrgnuti endokrinoj terapiji.
Pod ovim okolnostima, 48 pacijenata sa kancerom dojke dobijalo je 3 grama dnevno spora Reishi gljive. Kod njih su zabeležena značajna poboljšanja u fizičkom blagostanju i umoru, smanjenje anksioznosti i depresije, kao i poboljšanje kvaliteta života. Osim ovoga, markeri CRF bili su značajno niži.
The fatigue prevalence in breast cancer survivors is high during the endocrine treatment. However, there are few evidence-based interventions to manage this symptom. The aim of this study was to investigate the effectiveness of spore powder of Ganoderma lucidum for cancer-related fatigue in breast cancer patients undergoing endocrine therapy. Spore powder of Ganoderma lucidum is a kind of Basidiomycete which is a widely used traditional medicine in China. 48 breast cancer patients with cancer-related fatigue undergoing endocrine therapy were randomized into the experimental or control group. FACT-F, HADS, and EORTC QLQ-C30 questionnaires data were collected at baseline and 4 weeks after treatment. The concentrations of TNF-α, IL-6, and liver-kidney functions were measured before and after intervention. The experimental group showed statistically significant improvements in the domains of physical well-being and fatigue subscale after intervention. These patients also reported less anxiety and depression and better quality of life. Immune markers of CRF were significantly lower and no serious adverse effects occurred during the study. This pilot study suggests that spore powder of Ganoderma lucidum may have beneficial effects on cancer-related fatigue and quality of life in breast cancer patients undergoing endocrine therapy without any significant adverse effect.
Chaga (čaga)
Gljiva Čaga naširoko se koristi u narodnoj medicini Rusije i istočne Evrope za lečenje raka i kao podrška imunom sistemu. Zaslužna je za lečenje od raka čuvenog ruskog pisca Aleksandra Solženjicina.
Čaga sadrži visok nivo betulinske kiseline, koja je jedan od ključnih sastojaka imele iz kore drveća, na kojima se Čage i susreću “zalepljene”. Njene dokazane osnovne osobine su antiretroviralnost (sprečava ponovnu pojavu virusa), antimalaričnost, antiinflamatornost i antikancerogenost.
Betulinska kiselina i rak
Betulinska kiselina iz Čage indukuje mitohondrijsku apoptozu u različitim ćelijskim linijama kancera i inhibira enzim topoizomerazu, koja je neophodna za odmotavanje i namotavanje DNK lanaca u replikaciji ćelije.
Laboratorijske studije o betulinskoj kiselini pokazale su da je veoma efikasna protiv širokog spektra malignih ćelija: humani melanom, neuroblastom, meduloblastom, Juingov sarkom, tumori mozga, karcinom jajnika, leukemija…
Dejstvo betulinske kiseline protiv tumorskih ćelija mozga posebno je interesantno i važno je napomenuti da je u jednoj studiji izvršeno citotoksično dejstvo protiv primarnih ćelija kod pacijenata, i to 4 od 4 testirana uzorka meduloblastoma i kod 20 od 24 testirana uzorka glioblastoma.
Betulinska kiselina takođe pokazuje veliki potencijal u kombinaciji sa radioterapijom, pokazujući strogo aditivni način inhibicije rasta u kombinaciji sa zračenjem u ćelijama melanoma u jednoj studiji, dok u drugoj deluje kao radiosenzitajzer (maligne ćelije čini osetljivijima na radijaciju) skvamoznih ćelija raka koji se javlja u predelu glave i vrata.
New therapies using novel mechanisms to induce tumor cell death are needed with plants playing a crucial role as a source for potential anticancer compounds. One highly promising class of natural compounds are the triterpenoids with betulinic acid (BetA) as the most prominent representative. In-vitro studies have identified this agent as potently effective against a wide variety of cancer cells, also those derived from therapy-resistant and refractory tumors, whereas it has been found to be relatively nontoxic for healthy cells. In-vivo preclinically applied BetA showed some remarkable anticancer effects and a complete absence of systemic toxicity in rodents. BetA also cooperated with other therapies to induce tumor cell death and several potent derivatives have been discovered. Its antitumor activity has been related to its direct effects on mitochondria where it induces Bax/Bak-independent cytochrome-c release.
Cordyceps
Pored dobro poznate imuno-modulacije koju izazivaju jedinjenja polisaharida, postoje dokazi o još jednom mehanizmu kada je u pitanju dejstvo Cordycepsa na tumor. To je povezano sa strukturom nekih od izmenjenih nukleotida koje sadrži ova gljiva, a koje imamo u primeru jedinjenja kordicepina (3 dezoksiadenozina).
Detaljan mehanizam dejstva pogledati na https://www.moj-imunitet.com/cordyceps.html
Imunološka stimulacija na polisaharide iz gljiva
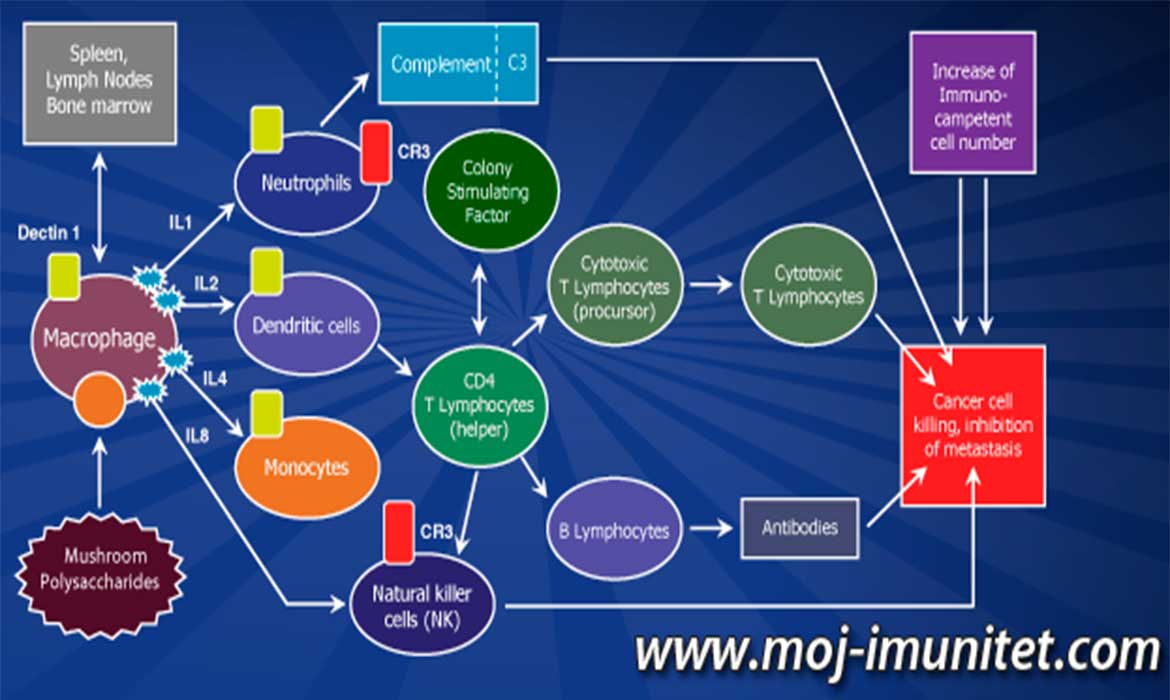
Putevi moduliranja imuniteta pomoću proteoglikana iz gljiva, odnosno način formiranja imunog odgovora na polisaharide gljiva prolazi kroz određene procese (vidi sliku). Aktivne komponente iz medicinskih gljiva deluju na imuni sistem tako što stimulišu stvaranje:
- Makrofaga, neutrofila, ćelija prirodnih ubica, dendričnih ćelija
- Povećane proizvodnje C3 komplemenata
- Uvećanog stimulišućeg faktora proizvodnje kolonija (Colony stimulating factor)
- Sinteze i otpuštanja interleukina: (IL)-1, IL-2, IL-4, IL-6, IL-8, IL-13, (TNF alfa), Interferoni (IFNs)
- Povećanja CD8 + T ćelija (Citotoksični T limfocit)
- Povećanja CD4 + T ćelija (pomoćničke T ćelije)
- Th1 > Aktivacija / Th2
- Prevencije metastaza – (Matriks Metaloproteinaze, MMPs)
- Indukcije apoptoze
- Inhibicije angiogeneze i invazivnosti tumora
- Imunomodulatornog efekta i antiinflamatorne aktivnosti
- Ublaženosti ili potpune otklonjenosti bola izazvanog kancerom

Gljive i hemoterapija
Lentinan iz gljive Shiitake je produžio opstanak i smanjio incidencu neželjenih efekata kod neoperativnog ili povratnog raka želuca u kombinaciji sa tegafurom kao i hemoterapiji zasnovanoj na S-1 (tegafur, gimeracil, oteracil).
Petogodišnje preživljavanje bez bolesti kod slučajeva kancera debelog creva značajno je poraslo uz PSK iz gljive Coriolus i oralno uzimanje Tegafura/Uracila (UFT), nego samo uzimanje UFT(73 % u odnosu na 58.8 %). Takođe, u slučajevima karcinoma želuca povećan je petogodišnji period bez bolesti (70,7% u odnosu na 59.4%) i preživljavanje (73,0% vs 60,0%) kada se PSK daje zajedno sa mitomicinom i fluoracilom.
Ekstrakti polisaharida iz gljive Agaricus blazei umanjili su propratne efekte hemoterapije (apetit, alopecija, emocionalna stabilnost i opšta slabost) u 100 slučajeva raka grlića materice i jajnika kod obolelih pacijenata koji su se lečili karboplatinom u kombinaciji sa etopozidom (VP16) ili taksolom.
Frakcije polisaharida iz gljive Maitake potencirale su akciju karmustina i povećale efikasnost kada se daje u kombinaciji sa hemoterapijom u većem broju kancera, smanjujući nefrotoksičnost indukovanu.
Gljive i radioterapija
Smanjenje broja leukocita tokom indukovane radijacije posmatrano je u ogledu sa 136 pacijenata koji su bili podvrgnuti radioterapiji. Konzumiranje ekstrakta polisaharida gljive Tremella fuciformis (3 grama dnevno) rezultirao je smanjenjem belih krvnih zrnaca od 13,2 % u odnosu na smanjenje od 35,2 % kod kontrolne grupe, dok je konzumacija ekstrakta gljive Coriolus versicolor (6 grama dnevno) sprečila pad crvenih i belih krvnih zrnaca kod bolesnika sa karcinomom pluća koji su podvrgnuti tretmanu radioterapije.
Poboljšan tretman efikasnosti kroz oralnu primenu ekstrakta polisaharid peptida (PSP) gljive Coriolus versicolor zajedno sa radioterapijom, značajno je povećao procenat apoptotičnih ćelija u 24 sata, u odnosu na rezultat samog zračenja, a takođe i smanjio redukciju leukocita koju je uzazvala radioterapija.
Zaključak
Proizvodi od medicinskih gljiva postepeno bivaju globalno priznati i na putu su da naprave revoluciju u terapeutskim strategijama u suzbijanju različitih oblika raka. Oni su efikasni i najvažnije
NISU TOKSIČNI !!!
Prevencija kancera i lečenje nekad mogu da budu jedno te isto!
Ljudi se smatraju “zdravima” sve dok ne pokažu simptome. Ako smo skrivali malignitet 20 godina, možemo se osećati dobro, ali to nismo.
Ako jednoga dana otpočnemo sa prevencijom raka, mogli bismo, baš u tom trenutku, da počnemo da ga lečimo, a da to uopšte i ne znamo. Upravo zbog ovoga, prevencija raka i njegovo lečenje ponekad mogu da budu jedno te isto!